Import Medical Cannabis to Europe
Home » Import Medical Cannabis to Europe
How to Import Medical Cannabis From Africa to Europe (2023)
- Last Update: March 2023
- By: WeGROW Team
There is considerable variation in the regulatory approach to import medical cannabis to Europe between the union members. Yet, despite the complexity, there is a clear route to import medical cannabis to Europe with WeGROW's registered high quality, rich THC & EU-GMP certified importing process.
Medical cannabis has shown efficacy in trials and is being deployed legally in medical settings as a therapeutic for conditions ranging from palliative care for cancer to appetite stimulant in patients with AIDS-related wasting syndrome, that’s why import medical cannabis to Europe will make it more accessible for more patients.
Across Europe, some countries allow the use of medicinal products containing cannabinoids, while others allow the medical use of unauthorized products or preparations. Some have standalone medical cannabis programmed. Some countries allow cannabis product manufacture. Others do not allow manufacture but do permit cannabis import, while in other countries you can do both.
Medical cannabis is still far too expensive, especially for out-of-pocket patients. Yet high quality cannabis can be grown very cheaply in many parts of the world like in Africa.
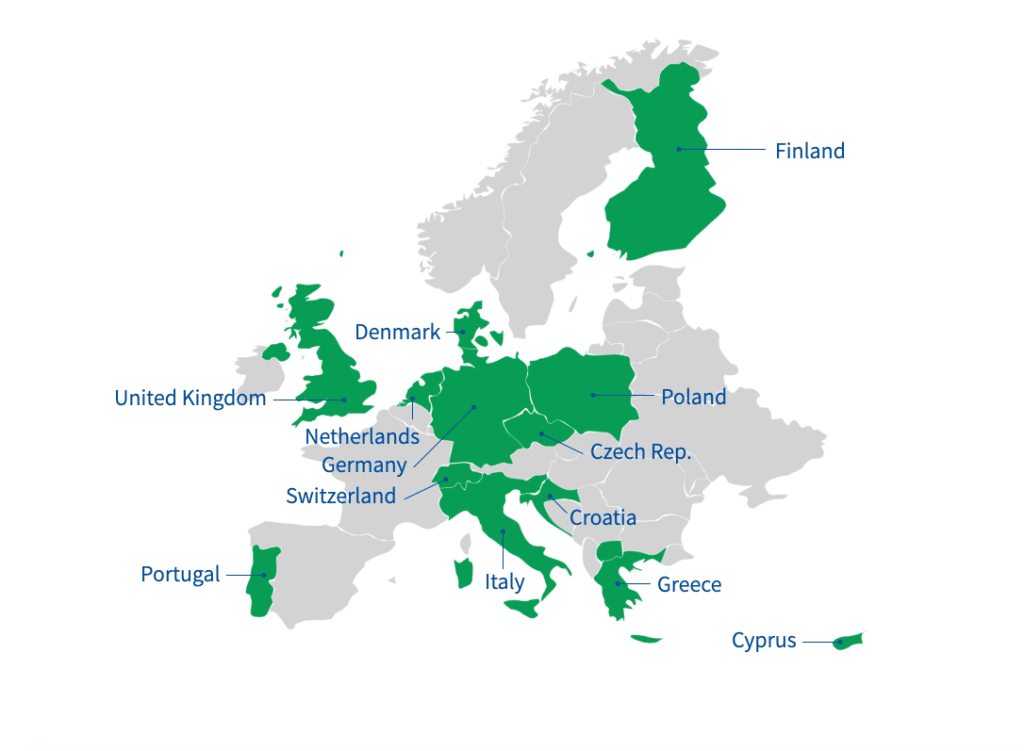
Legal Status of Importing Medical Cannabis Products into Europe
The first step in tackling that challenge is recognizing that European countries fall into three broad groups when it comes to their legal framework for medical cannabis. Some countries, including the UK, the Netherlands, Czech Republic, Poland, Switzerland and Portugal, have well-established medical cannabis legislation.
In many of these countries, the relevant legislation is quite recent. The government of Greece approved the legalization of cannabis for medical use as recently as 2018, the same year as the UK, while Germany made it legal for doctors to prescribe medical cannabis in 2017.
Cyprus is governed by the Medical Cannabis Law 2019, which states: “Patients will be supplied with medicinal cannabis by pharmacies upon presentation of a prescription from a specialist physician. This process ensures that patients will receive quality cannabis. under the supervision of a specialist, avoiding the need to obtain cannabis of dubious quality from the illegal market.”
In these countries the use of medical cannabis is clearly provided for under law and pharmacovigilance regulations relating to medical cannabis are the same as for medicinal products. That means patient safety is the same as for any other drug and the same measures can be taken to identify and deal with counterfeit drugs.
In other countries, including Sweden, Latvia, Belgium, and Albania, medical cannabis is strictly prohibited. It is illegal to use, buy or sell cannabis. At the present time, there are no routes to market medical cannabis into those countries.
Somewhere in the middle are countries that permit the supply of specific cannabis products under specific conditions. These include countries with trial period or pilots underway examining the possibilities for the use of medical cannabis. In these countries there are opportunities to market medical cannabis, with care.
Some countries allow import of medical THC cannabis at the discretion of physicians, usually relating to specific treatment for a named patient. In Croatia, for example, there is just one cannabis medicinal product registered containing cannabidiol (Epidyolex). The only way to register cannabis there is through an imported license.
In the UK, the medical cannabis industry is projected to be worth up to £2 billion by 2024. With an estimated 1.4 million patients eligible for treatment. As the industry grows, it has the potential to create new jobs, generate tax revenue, and stimulate economic growth by opening new import routes to the UK.
France, Ireland, and Denmark are running trial periods for medical cannabis, while in Finland only Sativex (an oromucosal spray) is available on prescription, for patients with MS. CBD may be prescribed for other conditions, decided on a case-by-case basis by the Finnish Medicines Agency (FIMEA). A prescription can only be issued by a neurological expert (or doctors in a neurological clinic) and is viewed as a ‘last-resort’ if the patient hasn’t responded to other medication.
While some countries have a specific regulations relating to medical cannabis, in others, products that contain cannabinoids that do not have any formal authorization as medicinal products, may have a narcotics license , as they do in Germany, for example. Similar narcotics legislations in other countries may provide life sciences companies with a route into these markets.
In European countries that do allow medical cannabis, whether in established law or in pilot projects, there is a further issue relating to the form of preparation allowed. Medical cannabis may be supplied in oils, capsules, as dried plants or a liquid solution. On top of that, there is a lack of standardization in the manufacturing of medical cannabis products across different markets. only one principle is consider the golden industry standard: EU-GMP.

Threshold of Importing for Medical Purposes: EU-GMP Certificate
A Good Manufacturing Practice (“GMP”) certificate is required in accordance with the rules governing medicinal products in the European Union (“EU”). The EU GMP certification is awarded by the German competent agency, the Authority for Medicines (GER: Arzneimittelbehörde).
EU-GMP standards for pharmaceuticals are among the most rigorous in the world, demanding the highest levels of quality assurance and product consistency. Usually following a pre-audit review by a local compliance Auditor, a comprehensive inspection is conducted over a 4-day period by government officials from the responsible agency on-site.
Medicines must have a clearly defined composition. This means the cannabinoid and terpene content is consistent from one batch to another.
Such consistency is important for:
- Doctors and their patients – to assure dosage composition and repeatability
- Researchers – to compare findings from clinical trials and studies across time
- Industry – to access GMP-certified active pharmaceutical ingredient, and therefore guarantee seamless GMP manufacturing
Importing Process of Cannabis Products Into Europe
Generally, there is not a single route for Import Medical Cannabis to Europe approval. Each country has established its own regulations regarding Cannabis, for example the import of high quality cannabis products for medical purposes from WeGROW Lesotho in Germany needs a permit in accordance with Section Three of the Narcotics Act (“BtMG”).
The main product-related prerequisite for the granting of narcotics permits for import. Is that the cannabis intended for import or used for the production of preparations is grown in the country of origin. Our colleagues at AFRICANN mentioned that for medical purposes under state control is in accordance with articles 23 and 28 of §1 of the single convention of 1961 on Narcotic Drugs. Evidence must be provided with the application.
In addition, the following questions must be clarified before the application is submitted, along with the relevant documents attached to the application:
Which varieties of cannabis flowers or which preparations made from cannabis should be traded? Product specifications must be submitted.
- Which country does the cannabis come from?
- From which company, from which country, and what annual maximum quantity should the cannabis flowers or preparations be obtained?
- If the necessary pharmaceutical permits have already been applied for the copies of these must be attached to the application.
- Be aware that an importer needs to provide all necessary state authorisation before an import license can be done.
- Licenses under the AMG (Medicines Act) are issued by the authorities responsible under State Law.
In Germany the basic requirement under the narcotics law for trading in medical cannabis is a license in accordance with Section three of the Narcotics Act (BtMg). In addition to this permit, an import permit in accordance with Section 11 BtMg of BfArm is required for each individual import of medical cannabis. After the necessary permit according to §3 BtMg has been received, the permit for each individual import can be applied for. The import permit necessary under the Medicinal Products Act (AMG) must be distinguished from the narcotics law permit. Learn more about Import Cannabis to Germany specifically.
Explore the differences between medical craft cannabis and mass-produced cannabis and learn about the benefits of crafting in Africa.

Benefits of Importing from WeGROW Lesotho
Pricing: Medical cannabis is still far too unaccessible and expensive, especially for out-of-pocket patients. Yet high quality cannabis can be grown very cheaply in many parts of the world like in Africa. We are bringing it to Europe. We guarantee highest quality through our production process and pass on the price advantages to patients through low production costs and high volumes.
Branding: WeGROW operates as a trusted long-term production partner offering white or private label services for the importer to build their brand in the markets they operate.
Market Fit: WeGROW fullfils the strict German and European quality standards for medicinal cannabis. As a result, patients receive cannabis flowers as high-quality medicinal products that meet all regulatory requirements according to the EU-GMP standard.
Logistics: Sending cannabis around the world is not that easy. In close coordination with WeGROW’s processing partners, they coordinate the obtaining of import and export permits and advise them on all issues that may arise – from the correct transport packaging to the choice of the logistics company. Once arrived in Germany, the cannabis gets prepared for production into a medicinal product in a highly secured and climatically standardised storage facilities.
Processing: The essential manufacturing steps for cannabis medicinal products take place in an EU GMP (Good Manufacturing Practice) certified production facility in Germany. Every single flower is processed and inspected. We do not use ionising radiation at all, which helps to protect the terpenes and active substances in the plant.
Quality: Protocols of controls and laboratory analyses at various points along the production process ensure the reliable pharmaceutical quality of the end product. Patients can thus concentrate fully on their therapy and do not have to worry about the quality of the medicines.
About WeGROW's High Quality Cannabis Production
WeGROW Cannabis in Lesotho has both its cultivation and processing facilities based 1,600 meters above sea-level in the mountains of Lesotho, where clean air and peaceful environment ensure the growth of high quality cannabis plants according to strict EU-GMP Principles for cultivation, extraction and the processing of cannabis for the medicinal and pharmaceutical markets by exporting medical cannabis to Europe and other global cannabis markets.
The 60,000 square meter cannabis facility has 30,000 square meter of production space in a unique hybrid greenhouse environment and employs >100 members of the surrounding communities.
The greenhouse is built to harness the power of the sun and improve the quality of the cannabis flower. The unique greenhouse design utilizes the power of the sun to increase terpenes and resin development throughout the life cycle of the plant. This unique growing technique, paired with innovative processing techniques, allows for a finely cured and polished product that consumer across Europe enjoy.
WeGROW’s products are reliable because each variety contains a consistent amount of cannabinoids and terpenes. They are also free of contaminants such as microbes, pesticides and heavy metals. These qualities make WeGROW cannabis products reliable and safe for patients, while ensuring distributors a safe route to import cannabis from Africa.
Energy Efficient
WeGROW is producing at highest quality in a facility that sets the gold standard for the cannabis industry. Our greenhouses are powered by sunlight to provide optimal growing conditions.
Accessible
Compassionate pricing is a game-changer that not only subscribes to our values of accessibility for all, but makes us highly competitive as a supplier to international markets.
About WeGROW's Organic Rich THC Medical Strains
The company has developed a unique genetic portfolio of 13 stable high demanded medical grade strains backed with years of successful reputation import medical cannabis to Europe. These strains are highly THC concentrated and specially adopted for chronicle pain syndromes.
Interested to Import Medicinal Cannabis to Europe?
Leave us a message and one of our Import/Export team members will get back to you ASAP
Share / Send
Quick Navigation
Subscribe for Updates
Share / Send
Frequently Asked Questions and Answers on how to import medical cannabis to Europe
What is medical cannabis and why is it imported to Europe?
Medical cannabis refers to cannabis or marijuana that is used for medical purposes, typically to relieve symptoms of certain medical conditions. The import of medical cannabis to Europe is necessary because not all European countries have legalized medical cannabis, and those that have often have limited domestic production capabilities. Importing medical cannabis allows patients in these countries to access medical cannabis products that they would not otherwise be able to obtain.
Which countries in Europe import medical cannabis?
Several countries in Europe import medical cannabis, including Germany, the Netherlands, Portugal and Denmark. These countries have established import frameworks to allow the import of medical cannabis products that have been authorized by regulatory authorities in other countries.
How is medical cannabis imported to Europe?
Medical cannabis is typically imported to Europe through licensed importers who have established relationships with cannabis producers in other countries. The imported cannabis products must meet certain standards for quality and safety and must be approved by regulatory authorities in the importing country.
What types of medical cannabis products are imported to Europe?
Medical cannabis products that are imported to Europe include dried cannabis flower, cannabis extracts, and cannabis-based pharmaceutical products. These products are used to treat a variety of medical conditions, including chronic pain, multiple sclerosis, and epilepsy.
What are the regulations governing the import of medical cannabis to Europe?
The regulations governing the import of medical cannabis to Europe vary by country. Generally, medical cannabis products must be approved by regulatory authorities in the importing country, and importers must have the necessary licenses and permits to import and distribute medical cannabis products. The products must also meet certain quality and safety standards and must be accompanied by appropriate documentation. Additionally, some countries may require that medical cannabis products be prescribed by a physician and dispensed by a licensed pharmacy.





