Import Cannabis
to Germany
Home » Import Cannabis to Germany
How to Import Cannabis From Africa to Germany (2023)
- Last Update: March 2023
- By: WeGROW Team
Import of Cannabis to Germany hit a new record of imported dry flowers for medical sales and scientific use in the first half of 2022, putting the European Union’s biggest market on pace to match or possibly surpass 2021’s record high total and become second biggest cannabis market to import into.
Import cannabis to Germany can currently only take place from those countries that have established a state cannabis agency and guarantee the production of medical cannabis under state control and EU-GMP standards, such as Denmark, the Netherlands, Australia and Lesotho.
Determination of the import quality is a central element. The quality of the imported cannabis is based on Good Manufacturing Practice (GMP) requirements as well as the certainty of the countries from which it is imported exercising strict state control to ensure pharmaceutical quality. The cultivation, processing and export of cannabis must be monitored by the government agency for canna-bis. The international cooperation of the Federal Office for Drugs and Medical Devices (BfArM) is based on the International Narcotics Control Board (INCB) convention, the cannabis authorities themselves internationally work together closely.

Status of Importing Cannabis Products into Germany
Imported quantities of medical cannabis have increased steadily since 2015. In 2015 and 2016, patients could only obtain medical cannabis with a special permit. Since the legalization of medical cannabis in 2017, the amount of imported canna-bis (dried flowers) was just under 1,000 kg, then increased to just under 8,000 kg in 2019. In 2020, there was another significant increase in the amount of imported dried flower needed for medicinal purposes to nearly 12 tons.
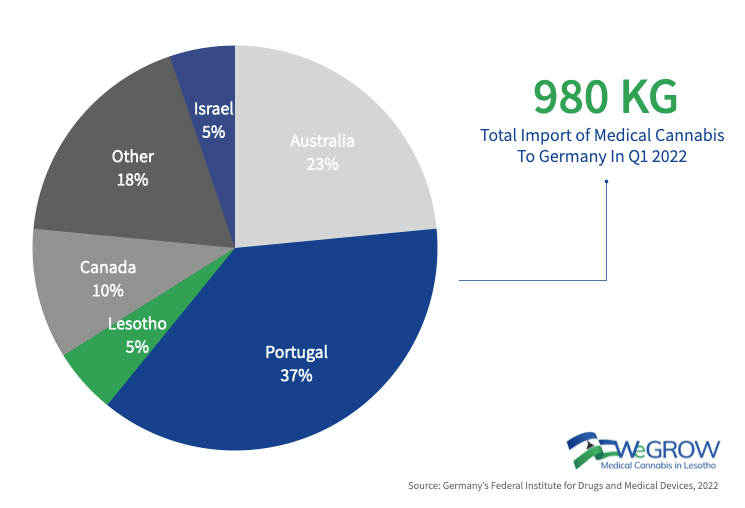
The following figure shows the quantities of medicinal cannabis flowers imported into Germany from abroad by country, mainly for dispensing to pharmacies for direct patient care:

In 2021, we have seen another significant increase in flower imports that was not expected. After four quarters, the volume of import cannabis to Germany (dried medicinal cannabis flowers) has now almost doubled compared to the previous year of 2020 = 11,746t, and of 2021 = 20,077t.
Import volumes for dried flowers in H1 2022 were 10,487kg, similar to H1 2021. However, it can be assumed that there will be further increases in import volumes in the next two quarters of 2022.
Reimbursement of Medical Cannabis Cost in Germany
Under the new German Medical Cannabis law, the cost of medical cannabis will be covered by health insurance providers, if
- The patient receives a valid prescription.
- The patient’s health insurance provider does not deny coverage, which is only possible in justified cases if the prerequisites of § 31 V Abs. 6 SGB V are not met.
- The prescribed cannabis is purchased at a licensed pharmacy (Apotheke) and meets all regulatory standards
.
The prescription of medical cannabis is at the discretion of the doctor. An official review of the prescription does not take place, or only in exceptional cases, when urgent indications of misuse of the prescription is implied, for example, if there is no indication at all. This is separate from the question of reimbursement, which is reviewed by the health insurers based on the diagnosis made.
Two thirds of these applications for reimbursement are approved, about one third are rejected. In Germany, about 90% of the population is compulsorily insured in the system of statutory health insurance.
It is estimated that over 100,000 patients are currently being treated with medical cannabis. The number of patients is not collected, only the number of prescriptions. Most available data refer to patients who have received reimbursement from their health insurance. No data are available for self-paying patients with statutory health insurance or self-paying private patients (approx. 10% of the population) without a reimbursement commitment.

It is therefore not possible to give an exact figure for the potential total sales volume of medical cannabis for the German market. The following figures only reflect the reimbursement of the statutory health insurance companies for non self-paying publicly insured patients. There are additional privately insured persons (about 10% of the population), whose data will not be collected, and also self-payers, whose application for reimbursement was rejected by the health insurance company or which haven’t applied for reimbursement at all:
In 2019, a total of € 122,995,551 in reimbursements for prescriptions containing med-ical cannabis was paid out by the statutory health insurance funds. Just under €54 million was reimbursed for unprocessed cannabis flowers, and just under €20 mil-lion for the finished drugs Canemes (€541,922) and Sativex (€18,258,020).
According to the German National Association of Statutory Health Insurance Funds (GKV-Spitzenverband), overall reimbursements of medical cannabis and cannabinoid-based medicines totaled 165.259.927,00 € in 2020 (Q1-Q4).
The published figures from January to September 2021 again show an increasing overall trend.
Cannabis flowers in unchanged condition were billed at € 53,453,323.00. From the figures, however, it is also clear that the reimbursement of unprocessed flowers by health insurance companies no longer shows significant growth rates.
The current growth we are seeing in dried flower imports is therefore likely due to private patients and self-pay patients who are not seeking reimbursement from health insurance companies. Currently, we can observe several business models that specialize in the low-threshold prescription of medical cannabis, such as Algea Care.
It is still expected that patient numbers in Germany as well as in Europe will increase continuously, after more and more patients and doctors have been made aware of this alternative form of therapy making in great business opportunity to begin import cannabis to Germany. Almost every country in the EU has already or will soon launch a medical Cannabis program, so that the pan-European demand will also increase continuously and develop into a veritable market. At this, individual regional, legal and cultural differences should be considered.
Threshold of Importing for Medical Purposes: EU-GMP Certificate
A Good Manufacturing Practice (“GMP”) certificate is required in accordance with the rules governing medicinal products in the European Union (“EU”) and to be able to import cannabis to Germany. The EU GMP certification is awarded by the German competent agency, the Authority for Medicines (GER: Arzneimittelbehörde).
EU-GMP standards for pharmaceuticals are among the most rigorous in the world, demanding the highest levels of quality assurance and product consistency. Usually following a pre-audit review by a local compliance Auditor, a comprehensive inspection is conducted over a 4-day period by government officials from the responsible agency on-site.
Medicines must have a clearly defined composition. This means the cannabinoid and terpene content is consistent from one batch to another.
Such consistency is important for:
- Doctors and their patients – to assure dosage composition and repeatability
- Researchers – to compare findings from clinical trials and studies across time
- Industry – to access GMP-certified active pharmaceutical ingredient, and therefore guarantee seamless GMP manufacturing
Process of Import Cannabis to Germany
The import of high quality cannabis products for medical purposes from WeGROW Lesotho to Germany needs a permit in accordance with Section Three of the Narcotics Act (“BtMG”).
The main product-related prerequisite for the granting of narcotics permits for import, is that the cannabis intended for import or used for the production of preparations is grown in the country of origin for medical purposes under state control is in accordance with articles 23 and 28 of §1 of the single convention of 1961 on Narcotic Drugs. Evidence must be provided with the application.
In addition, the following questions must be clarified before the application is submitted, along with the relevant documents attached to the application:
Which varieties of cannabis flowers or which preparations made from cannabis should be traded? Product specifications must be submitted.
- Which country does the cannabis come from?
- From which company, from which country, and what annual maximum quantity should the cannabis flowers or preparations be obtained?
- If the necessary pharmaceutical permits have already been applied for the copies of these must be attached to the application.
- Be aware that an importer needs to provide all necessary state authorisation before an import license can be done.
- Licences under the AMG (Medicines Act) are issued by the authorities responsible under State Law
.
In Germany the basic requirement under the narcotics law for trading in medical cannabis is a licence in accordance with Section three of the Narcotics Act (BtMg). In addition to this permit, an import permit in accordance with Section 11 BtMg of BfArm is required for each individual import of medical cannabis. After the necessary permit according to §3 BtMg has been received, the permit for each individual import can be applied for. The import permit necessary under the Medicinal Products Act (AMG) must be distinguished from the narcotics law permit.
All necessary documents required for permit application according to § 3 of the Narcotic Drugs Act (BtMG) can be found on the website of the Federal Opium Agency via the following link.
According to a public request by the German party “die LINKE”, the German Government answered further questions concerning medicinal cannabis in Germany. According to the government, by February 2021 almost 87 companies based in Germany hold a license to import medical Cannabis. The companies hold different types of licensing (e.g. import and/or manufacturing) for their companies and may be limited to a federal state (Bundesland, depending on the type of product and on the market release required.
In general, before you import cannabis to Germany there are four product categories you need to be aware of and are available under the reimbursement scheme: Cannabis flower, extracts, dronabinol (THC API) and off-label prescription for Sativex ® and Canemes®. CBD-rich products do not fall explicitly under the reimbursement but may be applied for cost coverage by the health insurance. Medical Cannabis and cannabinoid products for medicinal use are considered magistral preparations (the individual preparation of medicinal products based on a doctor’s prescription).
Current offerings are mainly in the high THC section. Cannabis flower and extracts with leveled THC:CBD ratio and high CBD variants have started to enter the market. Additional education offers have helped to establish a wider range of products and uses. Marketing and education activities directed to health care professionals are critical in respect to the number of prescriptions. The manufacturer or the whole-sale and distribution companies must offer even more information in order to open up further market potential.
Benefits of Importing from WeGROW Lesotho
Pricing: Medical cannabis is still far too unaccessible and expensive, especially for out-of-pocket patients. Yet high quality cannabis can be grown very cheaply in many parts of the world like in Africa. We are bringing it to Germany. We guarantee highest quality through our production process and pass on the price advantages to patients through low production costs and high volumes.
Branding: WeGROW operates as a trusted long-term production partner offering white or private label services for the importer to build their brand in the markets they operate.
Market Fit: WeGROW fullfils the strict German and European quality standards for medicinal cannabis. As a result, patients receive cannabis flowers as high-quality medicinal products that meet all regulatory requirements according to the EU-GMP standard.
Logistics: Sending cannabis around the world is not that easy. In close coordination with WeGROW’s processing partners, they coordinate the obtaining of import and export permits and advise them on all issues that may arise – from the correct transport packaging to the choice of the logistics company. Once arrived in Germany, the cannabis gets prepared for production into a medicinal product in a highly secured and climatically standardised storage facilities.
Processing: The essential manufacturing steps for cannabis medicinal products take place in an EU GMP (Good Manufacturing Practice) certified production facility in Germany. Every single flower is processed and inspected. We do not use ionising radiation at all, which helps to protect the terpenes and active substances in the plant.
Quality: Protocols of controls and laboratory analyses at various points along the production process ensure the reliable pharmaceutical quality of the end product. Patients can thus concentrate fully on their therapy and do not have to worry about the quality of the medicines.
About WeGROW's High Quality Cannabis Production
‘WeGROW’ has both its cultivation and processing facilities based 1,600 meters above sea-level in the mountains of Lesotho, where clean air and peaceful environment ensure the growth of high quality cannabis plants according to strict EU-GMP Principles for cultivation, extraction and the processing of cannabis for the medicinal and pharmaceutical markets by exporting medical cannabis to Germany and other global cannabis markets.
The 60,000 square meter cannabis facility in Lesotho has 30,000 square meter of production space in a unique hybrid greenhouse environment and employs >100 members of the surrounding communities.
The greenhouse is built to harness the power of the sun and improve the quality of the cannabis flower. The unique greenhouse design utilizes the power of the sun to increase terpenes and resin development throughout the life cycle of the plant. This unique growing technique, paired with innovative processing techniques, allows for a finely cured and polished product that consumer across Germany enjoy.
WeGROW’s products are reliable because each variety contains a consistent amount of cannabinoids and terpenes. They are also free of contaminants such as microbes, pesticides and heavy metals. These qualities make WeGROW cannabis products reliable and safe for patients, while ensuring distributors a safe route to import cannabis from Africa.
Energy Efficient
WeGROW is producing at highest quality in a facility that sets the gold standard for the cannabis industry. Our greenhouses are powered by sunlight to provide optimal growing conditions.
Accessible
Compassionate pricing is a game-changer that not only subscribes to our values of accessibility for all, but makes us highly competitive as a supplier to international markets.
About WeGROW's Organic Rich THC Medical Strains
As the leading Cannabis company in Lesotho we have developed a unique genetic portfolio of 13 stable high demanded medical grade strains backed with years of successful reputation import medical cannabis to Germany. These strains are highly THC concentrated and specially adopted for chronicle pain syndromes.
Interested in Importing Medicinal Cannabis to Germany?
Leave us a message and one of our Import/Export team members will get back to you ASAP
Share / Send
Quick Navigation
Subscribe for Updates
Share / Send
Frequently Asked Questions and Answers on how to import medical cannabis to Germany
Is it legal to import cannabis to Germany for medical purposes?
Yes, it is legal to import cannabis to Germany for medical purposes, as long as the import is authorized by the Federal Institute for Drugs and Medical Devices (BfArM). Patients who have been prescribed medical cannabis by a licensed physician can apply for an import license, and the cannabis must be obtained from a licensed producer in another country.
What are the requirements for importing medical cannabis to Germany?
To import cannabis to Germany for medical purposes, patients must have a valid prescription from a licensed physician, and the cannabis must be obtained from a licensed producer in another country. Patients must also apply for an import license from the BfArM, which requires them to provide detailed information about their medical condition, the prescribed dosage, and the proposed duration of treatment.
How much cannabis can be imported to Germany for medical purposes?
The amount of cannabis that can be imported to Germany for medical purposes depends on the approval of the BfArM. In general, companies are only allowed to import the amount of cannabis that is necessary for their patients, and they must provide evidence that the cannabis has been prescribed by a licensed physician in the market.
Can patients import cannabis for medical purposes on their own?
No, patients cannot import cannabis for medical purposes on their own, as the import must be authorized by the BfArM. Patients must apply for an import license and have the cannabis shipped directly to a licensed pharmacy in Germany. Additionally, patients must have a valid prescription from a licensed physician, and the cannabis must be obtained from a licensed producer in another country.
How long does it take to import cannabis to Germany?
The time it takes to import cannabis to Germany for medical purposes can vary depending on several factors, including the approval process by the BfArM, the shipping time, and the availability of the cannabis from the licensed producer. In general, importers should allow several weeks for the import process to be completed, and they should work closely with their customs and shipping partners to ensure that they have everything in place according to German regulations.





